 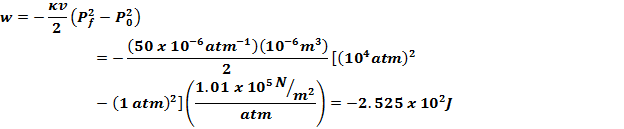 
This means that it is possible to use $dE=-P\,dV$ to calculate the change in the energy of the system. If you are applying a known pressure, in a nearly instantaneous compression, then there is essentially no time for the entropy of the system to change. In a irreversible compression, you can again calculate the work done, so long as you know a complete description of the system at all times. This is one instance in which you can calculate the work done by the gas (necessarily zero, because at each stage there is only free expansion) for a irreversible process moreover, for an ideal gas, with internal energy dependent only on temperature, not volume, it is easy to calculate the entropy generated by the expansion, showing it is irreversible. For example, a system may be allowed to expand freely in (infinitesimally) small increments for a gas, this would mean successively removing partitions, so that at each step there is only a minuscule increase in the volume of the gas, but with many iterations, the volume does increase by a finite amount. Any reversible process is necessarily quasistatic, but it is possible to have irreversible yet quasistatic evolution. This is a slightly more general condition than that the system needs to be evolving reversibly. 
However, this definition is only applicable when a system is undergoing quasistatic evolution, because that means that the system remains on (or infinitesimally close to) the equation of state surface, making it possible to take the derivative with respect to volume. $$P=-\left.\frac,$$įollowing from the expansion of the internal energy $E=U$ in its natural variables, There is a tendency among physicists when discussing thermodynamics to give a definition of pressure as That does not mean that it is impossible to do out-of-equilibrium calculations, but that it requires a different formulation of the dynamics in order to do those calculations. To a physicist, thermodynamics is essentially, by definition, the study of systems in thermal equilibrium. So, all I want to ask is: Why we don't do it like this in physics? What's wrong with it? Why do we say that we cannot calculate work done in irreversible processes like this? Please guide me towards the right path without using higher mathematics just explain it in brief, then it would be easier for me to understand. Let's assume $ W$ amount of work is done on the gas, so it means that it has increased the internal energy of the system by $ W$ (assuming no heat is released), so it also means that surely the work done by the system on the surroundings will be $-W$ to absorb that much energy, it has to do $-W$ amount of work on the surroundings, and there is no other thing from which energy can be exchanged. In chemistry for example, when we find the work done in irreversible compression, we can find how much work is done by the surroundings on the system because the pressure of the surroundings almost remains the same. We cannot directly find the work done in such a process, because we don't know what pressure of gas should we take, but I think, even if we cannot calculate this directly, we can calculate this indirectly like we do in chemistry. For example, the pressure in different regions of the system is different when a gas expands quickly. I am a high school student and I am very confused in thermodynamics, My confusion is that :In physics, when we study thermodynamics, we say that we cannot find the work done in irreversible processes because in such processes the different state variables of the gas are not defined. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
